Arginase (EC 3.5.3.1) belongs to the class of ureohydrolase. The systematic name of this enzyme is L-arginine amidinohydrolase. Other names in common use include arginine amidinase, canavanase, L-arginase, arginine transamidinase. Arginase is a manganese metalloenzyme that catalyzes the conversion of L-arginine to L-ornithine and urea. It was first identified in bacteria and later found in bacteria, yeast, plants, invertebrates and vertebrates. Most invertebrates, plants, bacteria and yeast have only one form of arginase, which is found in mitochondria, and vertebrate contains two forms of arginase, named A1 and A2. A1 is a cytosolic arginase and A2 is a mitochondrial arginase.
Structure
Human A1 (human arginase I) consists of 322 amino acids, while A2 (human arginase II) consists of 354 amino acids. These two isoforms are encoded by different genes on different chromosomes, but more than 50% of the amino acid residues are identical, with 100% homology in regions critical for enzyme function. The three-dimensional structure shows that both are trimers composed of three identical subunits and have an active site at the bottom of the 15 Å deep cleft. The manganese ions necessary for arginase activity are located at the bottom of the cleft. The overall folding of each subunit belongs to the α/β family, consisting of parallel, 8-stranded β-sheets flanked on both sides by a number of α-helices.
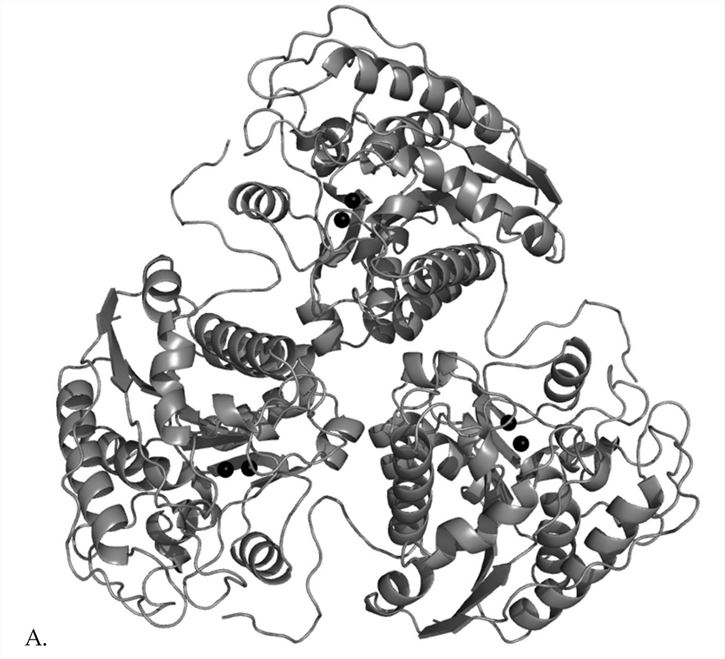 Figure 1. Structure of rat arginase trimer. (Dowling D.P. 2008)
Figure 1. Structure of rat arginase trimer. (Dowling D.P. 2008)
Amino acid side chains coordinated to the manganese ion in the arginase active site is located at the edge of the central β-sheet, in the loop segment adjacent to the strands 8, 7, and 4. There is a separation of MnA2+-MnB2+ in the binuclear cluster of unliganded arginase. The crystal structure of unliganded rat arginase I showed that MnA2+ was coordinated with H101, D128, 124, D232 and the hydroxide ion with square pyramid geometry. The crystal structure of unliganded human arginase I shows that MnA2+ coordinates with another water molecule to form a distorted octahedral metal coordination polyhedron. In rat arginase I and human arginase I, the MnB2+ is coordinated by H126, D234, D124, D232 and a hydroxide ion having a distorted octahedral geometry.
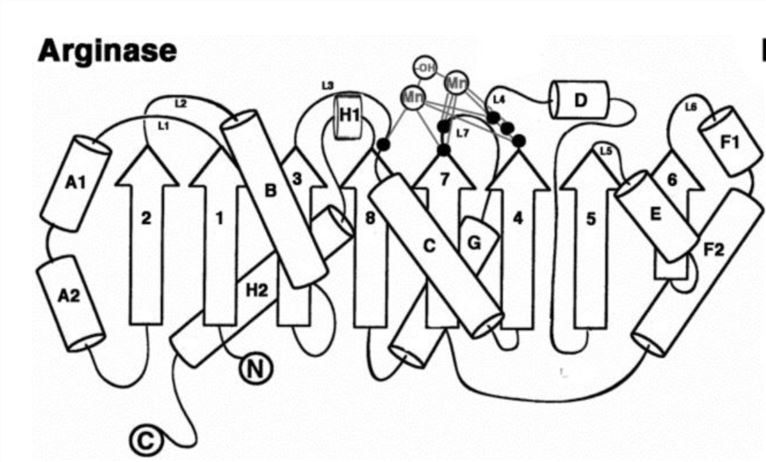 Figure 2. Secondary structure of arginase. (Dowling D.P. 2008)
Figure 2. Secondary structure of arginase. (Dowling D.P. 2008)
Catalytic Mechanism
In the human arginase I- 2(S)-amino-6-boronohexanoic acid (ABH) complex, ABH boronate hydroxyl O1 bridges MnA2+ and MnB2+ almost symmetrically. The ABH boronate hydroxyl O2 has a true inner-sphere coordination interaction. Since ABH undergoes the same chemical reaction with the substrate L-arginine at the arginase active site (nucleophilic attack by the metal-bridging hydroxide ion), and the boronate anion of ABH mimics the tetrahedral intermediate and its flanking transition states in catalysis, so the ABH boronate hydroxyl group O2 corresponds to the NH2 group of the substrate L-arginine. As the transition state approaches, MnA2+ interacts with the sp3 lone electron pair of NH2 groups through the interaction of strong inner-sphere bidentate coordination. Furthermore, the structure of the human arginase I-ABH complex provides an inference for the catalytic function of H141. The imidazole group of H141 acts as a proton shuttle to promote ionization of a metal-bridging water molecule, thereby regenerating nucleophilic metal-bridging hydroxide ions. The structure of human arginase I-ABH indicates that H141 forms a hydrogen bond with O2 of E277, and the carboxylate side chain of E277 also forms a weak hydrogen bond with the boronate hydroxyl group O2. Protons are present on the N atom of H141, and thus it is considered that H141 is stabilized by a positively charged imidazolium group by hydrogen bonding with E277. Then H141 is close to the -CH2 group of ABH, the -CH2 group is correspond to the leaving amino group of L-ornithine in the tetrahedral intermediate, and H141 can serve as a general acid catalyst to provide the proton for the leaving group departure. This function requires a 90° conformational change in the side chain torsion angle χ2 to determine the direction of H141 proton transfer, which also acidifies the imidazolium protons by destroying the E277 hydrogen bond. H141 imidazole is then used as a proton shuttle to regenerate the nucleophilic metal-bridging hydroxide ion in the final step of the catalysis.
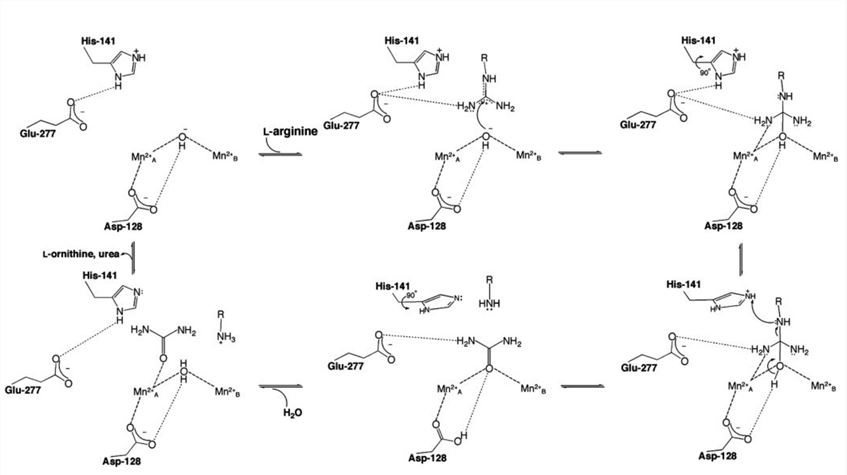 Figure 3. Mechanism of human arginase I. (Costanzo L.D. 2005)
Figure 3. Mechanism of human arginase I. (Costanzo L.D. 2005)
Application
Arginase activity has two main homeostatic purposes. The first is to remove ammonia from the body by urea synthesis, the second is to produce ornithine, which is a precursor of polyamines and prolines. Polyamines produced by ornithine decarboxylase (ODC) are required for cell proliferation and regulation of multiple ion channels. Proline produced by ornithine aminotransferase (OAT) is essential for collagen production.
Arginase plays an important role in health and disease, and it is involved in diseases and injuries affecting the peripheral cardiovascular system and the central nervous system (CNS). The expression and activity of arginase are up-regulated in many cardiovascular-related diseases such as hypertension, diabetes, and atherosclerosis. The expression of arginase is also up-regulated in a variety of CNS diseases such as stroke, Alzheimer's disease, Parkinson's syndrome and the like.
References
-
Caldwell, R.B., Toque, H.A., Narayanan, S.P., Caldwell, R.W. Arginase: an old enzyme with new tricks. Trends Pharmacol Sci, 2015, 36(6): 395–405.
-
Dowling, D.P., Costanzo, L.D., Gennadios, H.A., Christianson, D.W. Evolution of the arginase fold and functional diversity. Cell Mol Life Sci, 2008, 65(13): 2039–2055.
-
Costanzo, L.D., Sabio, G., Mora, A., Rodriguez, P.C., Ochoa, A.C., Centeno, F., Christianson, D.W. Crystal structure of human arginase I at 1.29-A resolution and exploration of inhibition in the immune response. Proc Natl Acad Sci U S A. 2005, 102(37):13058-63.


 Figure 1. Structure of rat arginase trimer. (Dowling D.P. 2008)
Figure 1. Structure of rat arginase trimer. (Dowling D.P. 2008)
 Figure 2. Secondary structure of arginase. (Dowling D.P. 2008)
Figure 2. Secondary structure of arginase. (Dowling D.P. 2008)
 Figure 3. Mechanism of human arginase I. (Costanzo L.D. 2005)
Figure 3. Mechanism of human arginase I. (Costanzo L.D. 2005)