Official Full Name
Phosphatase
Background
A phosphatase is an enzyme that removes a phosphate group from its substrate by hydrolysing phosphoric acid monoesters into a phosphate ion and a molecule with a free hydroxyl group (see dephosphorylation). This action is directly opposite to that of phosphorylases and kinases, which attach phosphate groups to their substrates by using energetic molecules like ATP. A common phosphatase in many organisms is alkaline phosphatase. Another large group of proteins present in archaea, bacteria, and eukaryote exhibits deoxyribonucleotide and ribonucleotide phosphatase or pyrophosphatase activities that catalyse the decomposition of dNTP/NTP into dNDP/NDP and a free phosphate ion or dNMP/NMP and a free pyrophosphate ion. The other group of phosphatase is collectively called as protein phosphatase, which removes a phosphate group from the phosphorylated amino acid residue of the substrate protein. Protein phosphorylation is a common posttranslational modification of protein catalyzed by protein kinases, and protein phosphatases reverse the effect.
Introductions
Phosphatase is an enzyme capable of dephosphorylating the corresponding substrate, i.e., removing the phosphate group from the substrate molecule by hydrolyzing the phosphate monoester and generating a phosphate ion and a free hydroxyl group. The role of phosphatases is the opposite of that of kinases, which are phosphorylases that can use energy molecules, such as ATP, to add phosphate groups to the corresponding substrate molecules. A phosphatase that is commonly found in many organisms is alkaline phosphatase.
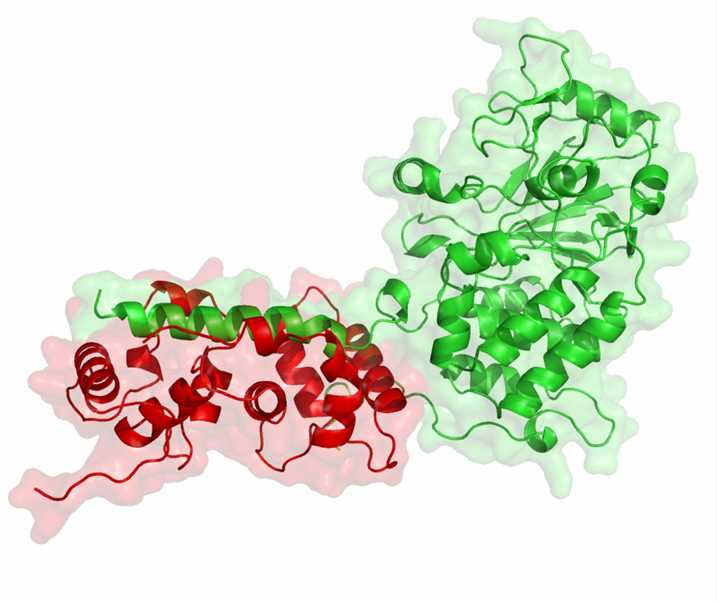 Figure 1. Structure of phosphatases.
Figure 1. Structure of phosphatases.
Acid phosphatase
Induction and secretion of acid phosphatase is one of the important adaptive responses of plants in response to low phosphorus environment. Acid phosphatases can hydrolyze phosphate groups from different organophosphorus substrates for plant uptake and utilization. Most plant acid phosphatases have no obvious substrate specificity and can hydrolyze substrates such as RNA, DNA, 3-phosphoglycerate, and hexose phosphate. Secretion of acid phosphatase is a common adaptive response to low phosphorus stress in plants, and this adaptive change is also the result of coordinated expression of phosphorus deficiency response genes, which promote phosphorus uptake, translocation and efficient utilization through the direct or indirect action of response gene products.
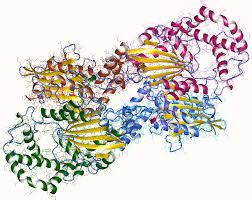 Figure 2. Acid phosphatase.
Figure 2. Acid phosphatase.
Alkaline phosphatase
Alkaline phosphatase is a non-specific phosphate monoesterase that catalyzes the hydrolysis of almost all phosphate monoesters to produce inorganic phosphate and corresponding alcohols, phenols, sugars, etc. It can also catalyze the transfer of phosphate groups and is a phosphite-dependent hydrogenase in E. coli A P. AP is present in almost all organisms except higher plants and can directly participate in phosphorus metabolism, playing a role in the digestion, absorption, secretion and ossification of calcium and phosphorus. It plays an important role in the digestion, absorption, and secretion of calcium and phosphorus, as well as in the process of ossification.
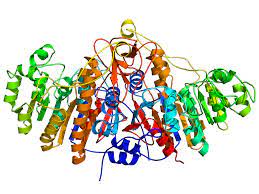 Figure 3. Alkaline phosphatase.
Figure 3. Alkaline phosphatase.
Biological function
Phosphatases are the opposite of kinases or phosphatases in terms of their phosphorylation. Phosphorylation can cause an enzyme to be activated or inactivated, or a protein-protein interaction can occur (e.g., SH2 structural domain). Thus, phosphatases are required for the control of phosphorylation by many signal transduction pathways. Phosphorylation or dephosphorylation does not necessarily correspond to activation or inhibition of the enzyme, and some enzymes have multiple phosphorylation sites involved in the regulation of activation or inhibition. For example, cyclin-dependent kinase (CDK) has multiple specific amino acid residues that can be phosphorylated, and activation or inhibition corresponds to phosphorylation of different residues. The reason why phosphate is important for signaling is its ability to regulate the action of the protein it binds, and the removal of phosphate is an inverse effect (if phosphorylation is activating, dephosphorylation is inhibiting), and it is here that phosphatases play an important role.
Catalytic mechanism
Cysteine-dependent phosphatases catalyze the breakage of phosphate bonds by forming a phosphate-cysteine intermediate in the following process (using the phosphorylation of tyrosine dephosphorylation as an example). First, the free cysteine nucleophilic group at the enzyme active site attacks the phosphorus atom in the phosphate group and forms a bond. Then, the P-O bond connecting the phosphate group to the tyrosine accepts a proton provided by a suitably positioned acidic amino acid (e.g., aspartic acid) or a water molecule and undergoes protonation, resulting in the formation of a phosphate-cysteine intermediate; this intermediate is hydrolyzed by another water molecule hydrolyzed, the active site of the enzyme is released and can proceed to the next dephosphorylation reaction. The active site of metal phosphatase combines two metal ions necessary for catalysis. The results of studies on the nature of these two metal ions are inconsistent and therefore inconclusive so far. The available evidence can only suggest that the two metals can be any combination of magnesium, manganese, iron, and zinc, and that the two metal ions are linked by a hydroxide ion. This hydroxide ion is currently thought to be involved in the nucleophilic attack on phosphorus.


 Figure 1. Structure of phosphatases.
Figure 1. Structure of phosphatases.
 Figure 2. Acid phosphatase.
Figure 2. Acid phosphatase.
 Figure 3. Alkaline phosphatase.
Figure 3. Alkaline phosphatase.