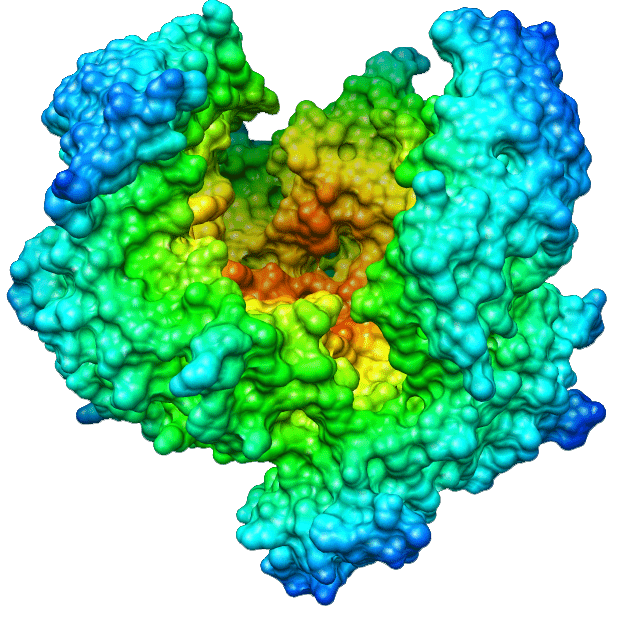
Streptokinase is a single chain polypeptide, which indirectly plays a fibrinolytic role by activating circulating plasminogen. The molecular weight of streptokinase is 47 kDa, which is composed of 414 amino acid residues. The protein showed the highest activity around pH7.5, and its isoelectric pH was 4.7. Proteins are free of cystine, cysteine, phosphorus, conjugated carbohydrates and lipids. Streptokinase (SK) is a thrombolytic drug and enzyme. As a drug, it is used to break down thrombus in some cases of myocardial infarction (heart attack), pulmonary embolism and arterial thromboembolism. The type of heart attack it uses is ST segment elevation myocardial infarction (STEMI). It's used by intravenous injection.
Structure
Streptokinase consists of multiple structural domains (i.e., a-, h- and g-domains) with different associated functional properties. Scanning calorimetric analysis suggests that the protein is composed of two distinct domains. The N-terminal domain (i.e., residues 1–59) has been found to complement the low plasminogen activation ability of the 60–414 amino acid residue domain of the protein.
Mechanism
Streptokinase belongs to a group of medications known as fibrinolytics, and complexes of streptokinase with human plasminogen can hydrolytically activate other unbound plasminogen by activating through bond cleavage to produce plasmin. Streptokinase is known to activate plasminogen both by fibrin-dependent and fibrinindependent mechanisms. Streptokinase interacts with plasminogen though multiple domains, including α (residues 1–150), β (residues 151–287), and γ (residues 288–414) domain. Each domain binds plasminogen, although none can activate plasminogen independently. At least two independent plasminogen binding sites of streptokinase had been identified. The C-terminal domain of streptokinase is involved in plasminogen substrate recognition and activation. Similarly, the Asp41–His48 region of streptokinase is important in binding to the substrate plasminogen.
Streptokinase binds preferentially to the extended conformation of plasminogen through the lysine binding site to trigger conformational activation of plasminogen. Streptokinase–plasminogen activator complex interacts with plasminogen through long range protein–protein interactions to maximize catalytic turnover. The first 59 amino acid residues seem to have multiple functional roles in streptokinase. Without these N-terminal residues, streptokinase has an unstable secondary structure. Loss of residues 1–59 greatly reduces the activity of the remaining streptokinase fragment (i.e., residues 60–414).
Enhancing Streptokinase
Immunogenicity of streptokinase and its relatively short half-life in circulation limit therapeutic potential of this protein. Streptokinase in circulation is proteolytically degraded by plasmin. Consequently, research has focussed on structurally modifying streptokinase to extend half-life, reduce or eliminate immunogenicity, and improve plasminogen activation. Mutant streptokinase with improved stability have been prepared. Two of the major sites of the proteolytic action of plasmin on streptokinase are Lys59 and Lys386. This knowledge has been used to engineer variants of streptokinase that are resistant to plasmin and, therefore, have a longer functional half-life. The plasmin-resistant forms of streptokinase have been found to be as active as the native streptokinase. Recombinant streptokinase produced in the yeast Pichia pastoris is glycosylated, and this appears to enhance its resistance to proteolysis. Following a similar path, attempts have been made to extend the half-life of native nonglycosylated streptokinase by complexing it with polymers such as polyethylene glycol (PEG).
Recombinant streptokinases with reduced immunogenicity have been produced. A mutant streptokinase that lacked the C-terminal 42 amino acids was found to be less immunogenic than the native molecule. One chemical modification has involved complexing streptokinase with PEG, primarily for reducing immunogenicity.
Medical Application
If percutaneous coronary intervention is not available within 90 to 120 minutes after first contact, streptokinase is recommended intravenously as soon as possible after (STEMI) the onset of a ST elevation myocardial infarction (STEMI). Because streptokinase is a bacterial product, the human body has the ability to enhance immunity to it. Therefore, it is recommended that the drug not be used again four days after the first administration, as it may be less effective and can cause allergic reactions. For this reason, it is usually given only for a person's first heart attack. Further thrombotic events could be treated with Tissue plasminogen activator (tPA). Overdose of streptokinase or tPA can be treated with aminocaproic acid.
Reference
-
Banerjee A, Chisti Y, Banerjee U C. Streptokinase—a clinically useful thrombolytic agent. Biotechnology advances, 2004, 22(4): 287-307.